Efficacy Results in Diagnostic Testing
Allow patients to remain euthyroid in diagnostic follow up using stimulated Tg Testing with or without a Whole-Body Scan
Comparing Thyrogen with THW in Tg Testing
Study Design
- In a prospective, randomized, phase 3 clinical trial with a cross-over, nonblinded design, 229 patients with well-differentiated thyroid cancer who had undergone total or near-total thyroidectomy were studied to compare whole body scans. Each patient was scanned first using THYROGEN, then scanned using thyroid hormone withdrawal. Tg levels were compared after Thyrogen to those at baseline and to those after thyroid hormone withdrawal (THW)3
- Patients who were included in the Tg analysis were those who had undergone total or near-total thyroidectomy with or without 131-I ablation, had < 1% uptake in the thyroid bed on a scan after thyroid hormone withdrawal, and did not have detectable anti-Tg antibodies.
- The maximum Thyrogen Tg value was obtained 72 hours after the final Thyrogen injection, and this value was used in the analysis.1

Stimulated Tg versus non stimulated Tg testing
Approximately 1 in 5 metastatic disease positives were missed with non stimulated Tg testing1
Among patients with metastatic disease confirmed by a post treatment scan or by lymph node biopsy (35 patients), Thyrogen Tg was positive (≥2.5 ng/mL) in all 35 patients, while Tg on thyroid hormone suppressive therapy was positive (>2.5 ng/mL) in 79% of these patients.1
As with THW, the intrapatient reproducibility of Thyrogen testing with regard to both Tg stimulation and radio iodine imaging has not been studied.
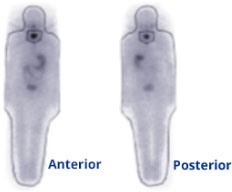
Uptake comparable in whole body scan (WBS)
Study Design
- Two prospective, randomized, phase clinical trials with a cross-over, nonblinded design, were conducted in patients with well differentiated thyroid cancer to compare 131-I WBS’s obtained after Thyrogen injection to 131-I WBS’s after THW. In both studies, the primary endpoint was the rate of comparable scan.1
- Across the two clinical studies the majority of positive scans using Thyrogen and THW were comparable.1
- The use of Thyrogen in preparation for WBS was comparable to THW in both efficacy and safety for stimulating the uptake of radioactive iodine (RAI).3
- Patients with residual thyroid tissue at risk for Thyrogen-induced hyperthyroidism include the elderly and those with a known history of heart disease.
WBS Image2
Choose how to get Thyrogen (thyrotropin alfa) for your patients
Not sure which one to choose? Contact ThyrogenONE for assistance

Option 1: Sign up or log into the ThyrogenONE portal
The ThyrogenONE portal provides your office online access to help you and your patients :
- Submit and track benefit verifications
- Coordinate orders
- Generate automatic alerts

Option 2: Call ThyrogenONE at 1-88-THYROGEN (1-888-497-6436) (
Dedicated ThyrogenONE Case Managers can assist you and your patients with the following.
- Benefits Verification
- Information on Ordering
- Information on specialty pharmacy fulfillment
Important Safety Information and Indications
Important Safety Information and Indications
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
If Thyrogen is administered with radioiodine, the contraindications to radioiodine also apply to this combination regimen. Refer to the radioiodine prescribing information for a list of contraindications for radioiodine.
WARNINGS AND PRECAUTIONS
Thyrogen-Induced Hyperthyroidism:
- There have been reports of death in non-thyroidectomized patients and in patients with distant metastatic thyroid cancer in which events leading to death occurred within 24 hours after administration of Thyrogen. Caution should be exercised in patients who have substantial thyroid tissue still in situ or functional thyroid cancer metastases, specifically in the elderly and those with a known history of heart disease.
- Hospitalization for administration of Thyrogen and post-administration observation in patients at risk should be considered.
Stroke:
- There are post marketing reports of stroke in young women with risk factors for stroke, and neurological findings suggestive of stroke (e.g., unilateral weakness) occurring within 72 hours of Thyrogen administration in patients without known central nervous system metastases. The relationship between Thyrogen administration and stroke is unknown. Patients should be well-hydrated prior to treatment with Thyrogen.
Sudden Rapid Tumor Enlargement:
- Sudden, rapid and painful enlargement of residual thyroid tissue or distant metastases can occur following treatment with Thyrogen. Pretreatment with glucocorticoids should be considered for patients in whom tumor expansion may compromise vital anatomic structures.
Risks Associated with Radioiodine Treatment:
- If Thyrogen is administered with radioiodine (RAI), the warnings and precautions for RAI apply to this combination regimen. Refer to the RAI prescribing information for a full list of the warnings and precautions for RAI.
ADVERSE REACTIONS
The most common adverse reactions reported in clinical trials were nausea and headache.
USE IN SPECIFIC POPULATIONS
Pregnancy:
- If Thyrogen is administered with radioiodine, the combination regimen is contraindicated in pregnant women.
- Available data with Thyrogen use in pregnant women are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Lactation:
- The concomitant use of Thyrogen and radioiodine (RAI) is contraindicated in lactating women. If Thyrogen is administered with RAI for diagnostic use, discontinue breastfeeding after RAI administration because of the potential for serious adverse reactions from RAI in the breastfed infant.
- If Thyrogen is not administered with RAI, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Thyrogen and any potential adverse effects on the breastfed child. There are no available data on the presence of thyrotropin alfa in human milk, the effects on the breastfed infant, or the effects on milk production.
Pediatric Use: Safety and effectiveness in pediatric patients have not been established.
Geriatric Use: Results from controlled trials do not indicate a difference in the safety and efficacy of Thyrogen between adult patients less than 65 years and those over 65 years of age.
Renal Impairment: Elimination of Thyrogen is significantly slower in dialysis-dependent end stage renal disease patients, resulting in prolonged elevation of TSH levels.
INDICATIONS AND USAGE
Thyrogen® (thyrotropin alfa) is a thyroid stimulating hormone indicated for:
Adjunctive Diagnostic Tool for Well-Differentiated Thyroid Cancer: Use as an adjunctive diagnostic tool for serum thyroglobulin (Tg) testing with or without radioiodine imaging in the follow-up of patients with well-differentiated thyroid cancer who have previously undergone thyroidectomy.
Limitations of Use:
- Thyrogen-stimulated Tg levels are generally lower than, and do not correlate with Tg levels after thyroid hormone withdrawal.
- Even when Thyrogen-Tg testing is performed in combination with radioiodine imaging, there remains a risk of missing a diagnosis of thyroid cancer or underestimating the extent of the disease.
- Anti-Tg Antibodies may confound the Tg assay and render Tg levels uninterpretable.
Adjunct for Thyroid Remnant Ablation in Well-Differentiated Thyroid Cancer: Use as an adjunctive treatment for radioiodine ablation of thyroid tissue remnants in patients who have undergone a near-total or total thyroidectomy for well-differentiated thyroid cancer and who do not have evidence of distant metastatic thyroid cancer.
Limitations of Use:
- The effect of Thyrogen on thyroid cancer recurrence greater than 5 years post-remnant ablation has not been evaluated.
Click here for full Prescribing Information
Important Safety Information and Indications
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
If Thyrogen is administered with radioiodine, the contraindications to radioiodine also apply to this combination regimen. Refer to the radioiodine prescribing information for a list of contraindications for radioiodine.
WARNINGS AND PRECAUTIONS
Thyrogen-Induced Hyperthyroidism:
- There have been reports of death in non-thyroidectomized patients and in patients with distant metastatic thyroid cancer in which events leading to death occurred within 24 hours after administration of Thyrogen. Caution should be exercised in patients who have substantial thyroid tissue still in situ or functional thyroid cancer metastases, specifically in the elderly and those with a known history of heart disease.
- Hospitalization for administration of Thyrogen and post-administration observation in patients at risk should be considered.
Stroke:
- There are post marketing reports of stroke in young women with risk factors for stroke, and neurological findings suggestive of stroke (e.g., unilateral weakness) occurring within 72 hours of Thyrogen administration in patients without known central nervous system metastases. The relationship between Thyrogen administration and stroke is unknown. Patients should be well-hydrated prior to treatment with Thyrogen.
Sudden Rapid Tumor Enlargement:
- Sudden, rapid and painful enlargement of residual thyroid tissue or distant metastases can occur following treatment with Thyrogen. Pretreatment with glucocorticoids should be considered for patients in whom tumor expansion may compromise vital anatomic structures.
Risks Associated with Radioiodine Treatment:
- If Thyrogen is administered with radioiodine (RAI), the warnings and precautions for RAI apply to this combination regimen. Refer to the RAI prescribing information for a full list of the warnings and precautions for RAI.
ADVERSE REACTIONS
The most common adverse reactions reported in clinical trials were nausea and headache.
USE IN SPECIFIC POPULATIONS
Pregnancy:
- If Thyrogen is administered with radioiodine, the combination regimen is contraindicated in pregnant women.
- Available data with Thyrogen use in pregnant women are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Lactation:
- The concomitant use of Thyrogen and radioiodine (RAI) is contraindicated in lactating women. If Thyrogen is administered with RAI for diagnostic use, discontinue breastfeeding after RAI administration because of the potential for serious adverse reactions from RAI in the breastfed infant.
- If Thyrogen is not administered with RAI, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Thyrogen and any potential adverse effects on the breastfed child. There are no available data on the presence of thyrotropin alfa in human milk, the effects on the breastfed infant, or the effects on milk production.
Pediatric Use: Safety and effectiveness in pediatric patients have not been established.
Geriatric Use: Results from controlled trials do not indicate a difference in the safety and efficacy of Thyrogen between adult patients less than 65 years and those over 65 years of age.
Renal Impairment: Elimination of Thyrogen is significantly slower in dialysis-dependent end stage renal disease patients, resulting in prolonged elevation of TSH levels.
INDICATIONS AND USAGE
Thyrogen® (thyrotropin alfa) is a thyroid stimulating hormone indicated for:
Adjunctive Diagnostic Tool for Well-Differentiated Thyroid Cancer: Use as an adjunctive diagnostic tool for serum thyroglobulin (Tg) testing with or without radioiodine imaging in the follow-up of patients with well-differentiated thyroid cancer who have previously undergone thyroidectomy.
Limitations of Use:
- Thyrogen-stimulated Tg levels are generally lower than, and do not correlate with Tg levels after thyroid hormone withdrawal.
- Even when Thyrogen-Tg testing is performed in combination with radioiodine imaging, there remains a risk of missing a diagnosis of thyroid cancer or underestimating the extent of the disease.
- Anti-Tg Antibodies may confound the Tg assay and render Tg levels uninterpretable.
Adjunct for Thyroid Remnant Ablation in Well-Differentiated Thyroid Cancer: Use as an adjunctive treatment for radioiodine ablation of thyroid tissue remnants in patients who have undergone a near-total or total thyroidectomy for well-differentiated thyroid cancer and who do not have evidence of distant metastatic thyroid cancer.
Limitations of Use:
- The effect of Thyrogen on thyroid cancer recurrence greater than 5 years post-remnant ablation has not been evaluated.
Click here for full Prescribing Information
References
1) Thyrogen Prescribing Information. Cambridge, MA: Genzyme Corp; 2020.
2) Hänscheid H, Lassmann M, Luster M, et al. Iodine biokinetics and dosimetry in radioiodine therapy of thyroid cancer: procedures and results of a prospective international controlled study of ablation after rhTSH or hormone withdrawal. J Nucl Med. 2006;47(4):648-654.
3) Haugen BR, Pacini F, Reiners C, et al. A comparison of recombinant human thyrotropin and thyroid hormone withdrawal for the detection of thyroid remnant or cancer. J Clin Endocrinol Metab. 1999;84(11):3877-3885.

.png)

.jpg 400w, /.imaging/webp/sanofi-platform/img-w500/dam/Thyrogen-hcp/Efficacy-in-Diagnostic-Testing/pagebanner--3-.jpg/jcr:content/pagebanner%20(3).jpg 500w)


